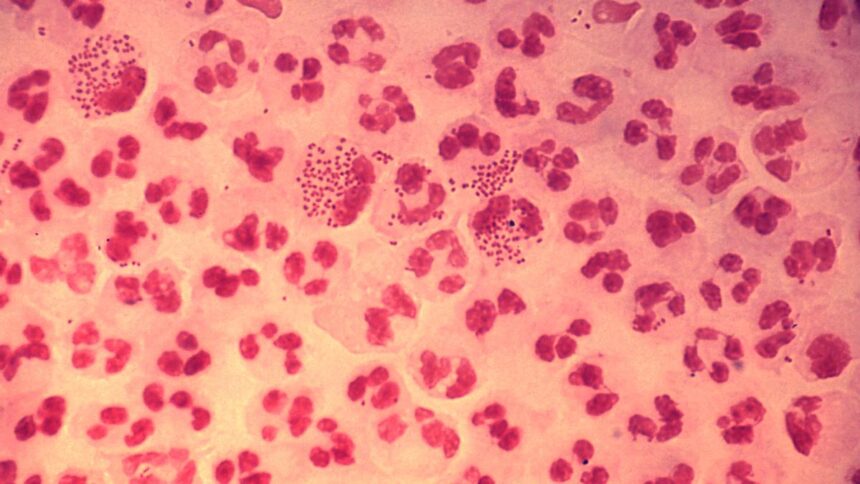
Neisseria gonorrhoeae, the bacterium that causes gonorrhea, is classed as a “excessive precedence” by the World Well being Group. The picture reveals the histopathology of an acute case of gonococcal urethritis. Photograph by: CDC/ Written by Joe Millar – This media is offered by the Facilities for Illness Management and Prevention’s Public Well being Picture Library https://commons.wikimedia.org/w/index.php?curid=722357.
The U.S. Meals and Drug Administration (FDA) has granted approval to 2 new oral medication to deal with gonorrhea, a standard sexually transmitted illness that’s vulnerable to drug resistance. Indian pharmaceutical large Dr. Reddy’s is taking steps to acquire advertising and marketing approval for one in every of these medication in Thailand and South Africa, and its unit, Origene Pharmaceutical Companies, will manufacture the drug.
The 2 medication, Nuzobenz (zoliflodacin), a water-soluble granule, and Bourjepa (gepotidacin), an oral pill, add vital parts to the arsenal in a world the place remedy choices for gonorrhea are quickly shrinking. “These approvals symbolize an vital milestone for remedy choices for sufferers with uncomplicated genitourinary gonorrhea,” Adam Sherwat, director of the Workplace of Infectious Illnesses within the FDA’s Middle for Drug Analysis and Analysis, stated within the launch. FDA approval for Nozolvence was given to Entasis Therapeutics, and approval for Blujepa was given to GSK.
Blujepa was developed by GSK, whereas Nuzolvence was a undertaking began by AstraZeneca however canceled and later taken over by Entasis, which partnered with the non-profit World Antibiotic Analysis and Growth Partnership (GARDP) and commenced trials in 2019.
“Excessive Precedence” Pathogens
The World Well being Group (WHO) maintains a listing of essential pathogens with an emphasis on antibiotic-resistant micro organism. The 2024 record consists of the micro organism that causes gonorrhea gonorrheais classed as “excessive precedence”. The reason being that they have an inclination to evade the consequences of medicine and are more and more immune to single medication and drug combos used as therapies all over the world.
Gonorrhea is unfold by means of sexual contact and normally impacts the throat, rectum, genitals, and eyes, and might trigger painful urination, bleeding within the genital space, and discharge. Many contaminated folks, particularly girls, are asymptomatic and sometimes go untreated, however untreated it could result in infertility and issues.
Instances are on the rise
In 2020, WHO estimated the variety of new infections amongst adults aged 15 to 49 to be 82.4 million. One of many causes that is growing is as a result of revolution within the area of HIV/AIDS remedy, explains Pierre Damard, GARDP’s analysis and improvement medicines and therapeutics undertaking chief. “Youthful generations have stopped utilizing condoms as a result of they’re not afraid of HIV. They’re utilizing pre-exposure prophylaxis (PrEP), a drug taken by folks vulnerable to contracting HIV to stop intercourse or injecting drug use. However PrEP doesn’t stop the transmission of sexually transmitted illnesses. There’s already proof that circumstances of gonorrhea are on the rise. There have been few new circumstances throughout the pandemic lockdown, however it seems to have exploded quickly after. ”
As a key a part of GARDP’s STI technique, Dr. Durham says zoliflodacin stays the one remedy for gonorrhea (Bourjepa can be used to deal with urinary tract infections). “Limiting scientific use on this approach ought to help acceptable stewardship of the drug and assist sluggish the emergence of resistance, thereby extending the efficacy of this new drug.”
Partnership with authorities
The drug is handy for sufferers as it’s administered in a single oral dose and will likely be launched first in Thailand and South Africa, the place scientific trials have additionally been performed. GARDP will even discover partnerships with governments to make the drug simply obtainable in these nations, Dr. Durham added.